PREVENTING ELECTROLYSIS
Electrolytic Corrosion (Electrolysis) occurs when dissimilar metals are in contact in the presence of an electrolyte, such as water (moisture) containing very small amount of acid. The dissimilar metals set up a galvanic action those results in the deterioration of one of them. The following is a list of the more common commercial metals, sequenced according to what is known as the “Galvanic Series”.
THE GALVANIC SERIES
1- Magnesium
2- Zinc
3- Aluminum
4- Iron
5- Steel
6- Nickel
7- Stainless Steel 400 Series
8- Tin
9- Lead
10- Brass
11- Copper
12- Bronze
13- Stainless Steel 300 Series
14- Silver
15- Gold
When any two metals in this list are in contact, with an electrolytic present, the one with lower number is corroded. The galvanic action increases as the metals are farther apart in the Galvanic Series. It is not always true that there is greater corrosion the down the scale one goes. In certain cases one metal immediately following another may be very corrosive.
One of most important facts that architecture should know about a metal or an alloy is its reaction with other metals or alloys with which it may be in contact. This data is given in Galvanic Series. Here the metals are listed in a sequence in which each metal is corroded by all that follow it. In other words, when two different metals are in contact with each other in the presence of moisture, there will be a flow of current from one metal (the “anode”(+)) to the other metal (the “cathode”(-)), and one will be eaten away, or disintegrated , while the other (the “cathode”) will remain intact.
An important point to remember in utilizing the Galvanic Series is that chief problem in this type of corrosion, and moisture depends a great deal upon climate. In the desert, electrochemical action will be at a minimum. On the seacoast the action will be much greater, not only because of the ever-present moisture, but also because of the salt. Some means of separating dissimilar metals must therefore always be found.
A copper roof should not be applied directly over a wood roof deck fastened with steel nails. Also, aluminum sheets should not rest directly steel purlins, but should be insulated from the steel with felt strips.
In any galvanic combination, the relative areas of the two materials forming the couple have a very important bearing on the extent of the corrosion. Under many conditions the extent of galvanic effect will be directly proportional to ratio of the area of the metal lower in the list.
Thus, if a piece of steel having a surface area of two square inches is coupled to a copper having a surface area of 100 square inches, the galvanic corrosion is roughly 50 times faster than it would be if the same piece of steel were coupled to piece of copper having a surface area of two square inches.
For this reason, it is wise to avoid galvanic couples where the exposed area of the metal lower in the galvanic series is much greater than that of the metal high in this series. As a practical example, it would definitely be dangerous to use a steel rivet to a copper plate, but reasonably safe to use a copper rivet to a steel plate. As far as possible, nails or other fasteners use in securing metal roof covings should be of the same metal as the roofing.
The two major
factors affecting the severity of galvanic corrosion are:
(1) the voltage difference between the two metals on the Galvanic Series (metals are farther apart in the Galvanic Series), and
(2) the size of the exposed area of cathodic metal relative to that of the
anodic metal.
Corrosion of the anodic metal is both more rapid and more damaging as the voltage difference increases and as the cathode area increases relative to the anode area.
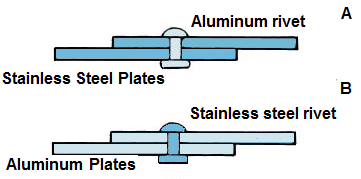
The effect of the second factor
above, the cathode-to-anode area ratio, C/A, is illustrated in above figure for
riveted plates. In both couples A and B, aluminum is the anode, and stainless
steel is the cathode. In couple A, the aluminum rivet is comparatively small,
and the C/A ratio is large. In couple B, the situation is reversed: the stainless
steel rivet is small, and the C/A ratio is also small. Corrosion of the
aluminum rivet in couple A will be severe. However, corrosion of the large
aluminum plate in couple B will be much less, even though the potential
difference is the same in each case.
What can you do to minimize galvanic corrosion? First, always try to eliminate the cathodic metal by making all parts of a structure out of the same material. When this is not possible, use nonmetallic, nonabsorbent insulators between the dissimilar metals to prevent current flow. For example, use plastic or ceramic washers and sleeves to isolate bolts as they pass through a plate of a different alloy instead of fiber and paper washers, which absorb water. Another approach is to make small critical parts out of the more cathodic metal so they will be protected. Always avoid connecting small anodes to large cathodes. Painting a galvanic couple can also be helpful in reducing the corrosion rate, but always be careful to paint both the anodic and cathodic members of the couple to keep the exposed C/A ratio small. If only the corroding member (the aluminum rivet) of couple A were painted, there would still be a large, bare cathode, which would make corrosion of the rivet even worse if the paint coating were scratched. That galvanic corrosion can be stopped by connecting both metals to a third metal more anodic than either of them. According to our Galvanic Series, the third metal in this case could be magnesium, zinc, aluminum, or cadmium. In practice, and for reasons too complex to cover here, zinc works best. (Some of the difficulties and dangers inherent in using the other anode materials are discussed in publications listed below.) The zinc corrodes preferentially to both of the original members of the couple. The steel is now protected, and the zinc is called a sacrificial anode. Such anodes are commonly used together with coatings to control corrosion on the underwater portions of boats, ships, and other marine structures. The same principle can be used to protect steel in marine atmospheres if the anodic metal is applied to the steel as a coating. Zinc (called galvanizing) and aluminum coatings are used extensively to protect steel in marine atmospheres.